The global pharmaceuticals market is forecast to grow to over $1 trillion by 2022 driven by aging demographics in developed countries and rising wealth levels in developing countries. An increasing portion of this market is expected to be biologics, such as those used in gene therapy. Given these growth trends, the losses can be expected to rise as well unless preventive logistic processes are implemented to protect the goods during both shipping and storing. Improving the existing supply chains to ensure a focus on precise temperature monitoring, cold chain monitoring, and data logging can help you preserve the integrity of your product, save money and provide better treatments for patients.
In 2017, the cold-chain logistics spending was globally over $13 billion for biopharmaceutical goods alone. Of the products that are included in that estimate, 20% to 40% are damaged before they reach the end-user. With the global spending in the industry being in constant rise, the total amount of pharmaceutical losses worldwide is immense.
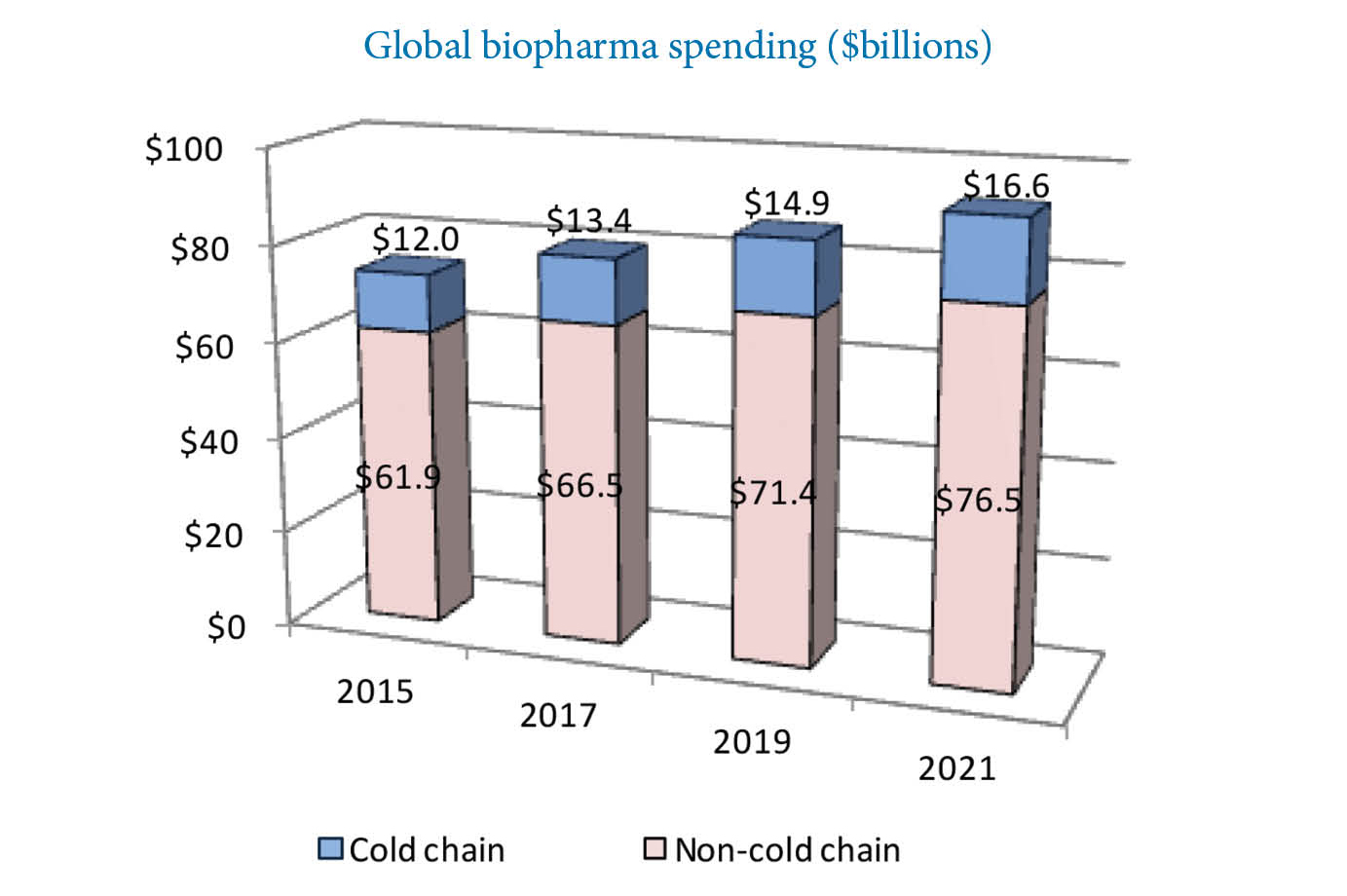 Global biopharma spending is continuously on the rise. Source: Pharmaceutical Commerce
Global biopharma spending is continuously on the rise. Source: Pharmaceutical Commerce
The typical online pharmacy process
Here’s what typically happens when a patient receives pharmaceuticals through an online pharmacy:
- They go to the doctor for an examination and receive a prescription
- They fill the prescription through an online pharmacy
- They receive the medication in the mail from a licensed pharmacy, with pharmacists available to answer questions about the purchase
The part that’s not seen by end-customers is the whole supply chain process that happens between filling and receiving the prescription: the online inventory has to be managed (including being protected and checked for safety standards) in order to provide satisfactory customer experience. That means anyone who owns an online pharmacy needs to be adept at working with suppliers and carriers. And they should also be invested in monitoring and reporting of data for their pharmaceutical supply.
Unfortunately, as I said in the intro, much of the supply can be at risk during the shipping process.
Problem: the growing complexity of the healthcare industry
Pharmaceutical products are sensitive or vulnerable to a wider range of circumstances than the products of almost any other industry. Biologics, samples from clinical trials and many classes of medications and medical products are extremely sensitive to heat, humidity and even light. On the other hand, many chemicals are also sensitive to vibrations and shocks.
While these items have life-saving capabilities, they are more complicated to research, develop, transport and store due to each and every product having to be as close to perfect as possible. No one wants their medication to have potentially fatal side effects.
 To maintain their effectiveness, different raw materials and finished goods may need to be stored at a variety of temperatures. Any disruption in this temperature or incorrect handling of the product could spoil it permanently. This could result in vaccines that are ineffective, pharmaceuticals and other temperature-sensitive products that must be thrown away or clinical trials that lose valuable data. Beyond this loss, you could also be subject to reputation risk and regulatory action.
To maintain their effectiveness, different raw materials and finished goods may need to be stored at a variety of temperatures. Any disruption in this temperature or incorrect handling of the product could spoil it permanently. This could result in vaccines that are ineffective, pharmaceuticals and other temperature-sensitive products that must be thrown away or clinical trials that lose valuable data. Beyond this loss, you could also be subject to reputation risk and regulatory action.
Is damage during shipping really that much of an issue? It absolutely is. Even if the number were relatively small, every item that cannot reach its destination intact is an inefficiency. But the number is not small. It’s clear that you need a system that can adapt to the evolving challenges in the healthcare industry.
According to an article in the journal Pharmacy and Therapeutics, the exact number of damaged pharmaceutical goods is difficult to pinpoint. That’s because the real number would include products that reach the end-user but are less effective than they should be. Depending on who and how makes the estimation, the numbers are bound to vary. However, some sources say the number to be at least 20 percent, and likely higher.
Losing over a fifth of any supply shouldn’t be accepted, from both financial and environmental standpoints. At that point, it’d probably be better to just have 20% less business in general, since you would at least save the production, shipping and storing costs.
A wide variety of factors can lead to damage. Among those are:
- Dropped packaging during picking, packing, shipping, and receiving
- Exposure to temperatures outside the safe range. And this doesn’t just mean keeping drugs cold. Some goods require room temperature conditions. Some require refrigeration. Others need to be frozen. And many cannot be allowed to freeze
- Exposure to light, which can degrade some products
- Incorrect humidity settings in the freight compartment, for humidity sensitive goods
- Shocks and vibrations during transportation that may cause packaging to tilt or fall over
Solution: implement or improve cold chain logistics
Whether your medical products require constant room temperature or need to be cooled, frozen or deep frozen, implementing cold chain logistics can help ensure temperature monitoring and management at every step of the process. Cold chain logistics refers to managing a supply chain for goods that must be kept at precise temperatures and/or humidity levels. While this has long been prevalent in the food industry, with refrigerated trucks and store rooms for perishable items, this type of supply chain is important in the healthcare field as well.
 Cold chain regulatory requirements vary by region. In the EU, for example, all medication packaging is required to come with a unique serial number and identifier. Given the complex regulatory landscape – with guidelines coming down from the EU Guide to Good Manufacturing Practice, CDC Guidelines for Maintaining and Managing the Vaccine Cold Chain, and US Code of Federal Regulations – it’s important to check with your governing body to ensure your cold chain procedures comply.
Cold chain regulatory requirements vary by region. In the EU, for example, all medication packaging is required to come with a unique serial number and identifier. Given the complex regulatory landscape – with guidelines coming down from the EU Guide to Good Manufacturing Practice, CDC Guidelines for Maintaining and Managing the Vaccine Cold Chain, and US Code of Federal Regulations – it’s important to check with your governing body to ensure your cold chain procedures comply.
The different aspects that need to be considered by the manufacturer, retailers, and sometimes even the end-customer, include:
- Storage and handling methods for refrigerated and frozen vaccines
- Considerations for equipment such as storage units and thermometers
- Strategies for maintaining the cold chain
- Routine storage and handling practices
- Inventory management and emergency procedures for protecting vaccine inventories (Source: VBI Vaccines) The different guidelines set by these different authorities exist to ensure that the pharma goods are safe to use - and effective. Any pharmacy can be in real trouble from not properly monitoring freight. The World Health Organisation and the EU Commission have strict requirements regarding the handling of pharmaceutical goods during shipping and storing.
The role of data logging
As the healthcare industry continues to evolve, technological advances will need to spread beyond the R&D phase to make sure manufacturing and distribution can keep up with the cutting-edge nature of these products. Cold chain solutions which incorporate temperature monitoring and data logging have tremendous potential for preserving the integrity of these products, reducing waste, saving money and improving health and safety.
The best way to ensure the safe arrival of pharmacy freight is to implement monitoring as part of the shipping process. Here is exactly why data logging is important for online pharmacy processes.
Important components include:
- Data logging devices that capture informatics such as temperature, vibration, humidity, etc.
- Devices placed in manufacturing plants, store rooms, transportation vehicles or with the product to provide temperature monitoring and other readings
- Advanced devices with GPS and RFID capabilities to provide real-time data updates
- Light sensors placed inside packages to verify the product hasn’t been tampered with
- Software that records and analyses all the information from the data loggers
- Packaging with built in temperature monitoring that can actively adjust the temperature of the items as needed
 How can data logging help any of these problems? It’s simple. When you monitor the conditions at which pharmaceutical goods are being transported, you can keep a record of how often appropriate conditions are being met, and when they are not. Once you have that information, you can then use it to prevent future incidents and ensure better shipping conditions.
How can data logging help any of these problems? It’s simple. When you monitor the conditions at which pharmaceutical goods are being transported, you can keep a record of how often appropriate conditions are being met, and when they are not. Once you have that information, you can then use it to prevent future incidents and ensure better shipping conditions.
In addition to actually spoilt goods, many products become too dangerous to use when spoilt. Because of this, even a suspicion of such a product being unusable leads to destroying it, simply due to the risks posed by the lack of knowledge whether it is safe to use or not.
Now, let’s assume you could determine with higher precision whether a specific part of your shipment is still usable. Simply increasing the monitoring precision from one measuring point to two increases the chances you can determine only a specific portion of the shipment unusable. For example, what if only half of the contents of your ruined shipments are actually ruined by average? That assumed 20% loss is instantly halved.
In a shipping operation worth a million a year, that could mean saving up to a 100k saved by a simple investment. The more precisely you can prove that a certain product is usable, the smaller the likelihood that you lose that product to mishandling. Even a simple, visible sign of implemented monitoring encourages proper handling of the packages.
Here’s how it works: - You install one or multiple sensors that record data input. Each sensor is designed to collect particular types of information. For instance, they may record sound, temperature, light, humidity, etc. These are usually battery operated, though you can also install sensors with external power connectors.
- The sensor device, or data logger, will also have a microprocessor and memory, in order to store the data, which can be transferred to a computer, or more likely now, transferred to cloud software so the information can be monitored and utilised right away. The speed of cloud computing has greatly improved the online pharmacy process with real-time monitoring.
- Supply chain managers check reporting status and analyse the data that has been collected, in order to improve supply chain strategies, taking each sensitivity into consideration.
But is monitoring really necessary?
 Yes, as I’ve pretty much established by now, monitoring your supply chain has real and immediate effects. For one thing, remember that estimates show at least 20% of pharmaceutical freight is being damaged during transport. That translates into real financial losses for your company. That 20% damage could mean:
Yes, as I’ve pretty much established by now, monitoring your supply chain has real and immediate effects. For one thing, remember that estimates show at least 20% of pharmaceutical freight is being damaged during transport. That translates into real financial losses for your company. That 20% damage could mean:
- Millions of dollars in lost supply
- Company reputation suffering from products arriving to customers in subpar condition
- Decreasing trust from consumers
- Environmental harm from disposed goods and waste
- Legal repercussions
Quality assurance increases trust
So, reputation and end customer trust. An important part of running a successful online pharmacy is strong brand presence and a positive perception by the public. Given that the pharmaceutical industry is in the business of helping people be healthier, it is imperative that the products arrive in top condition. Being aware of every part of the supply chain is the only way to know that you can guarantee the quality of your products.
When you use a suitable data logger to monitor what your products go through during shipping and storing, you can not only be certain of the validity of your goods yourself, but also can also show the data to any authorities asking questions about your operations, and even to the end-users if the logger is exceptionally efficient (Logmore and Finnish pharmacies are running a pilot project, in which the data loggers are attached to the product until they reach the end-user - quality assurance to the very end!). The trust attained with transparent operations is what earns your company the credibility that secures you new partners and customers - and a good reputation all around.
There are different types of data loggers available, but for the purposes of pharmaceutical transport, it’s best not to use standalone loggers that must have data manually transferred to a computer. That’s time-consuming and inefficient. Instead, a wireless logger connected to a cloud service is the best way to go. It can provide speedy data uploads for instant visibility to everyone who needs access to the data.

In conclusion
Efficient data logging is vital to any cold chain dependent operations, or any temperature-sensitive logistics in general. While the price tag may sound like an additional expenditure at first, simply being sure of which products can actually be used and which not can lead to your bottom line increasing by a lot just because you get to minimize the amount of products you have to consider spoilt due to uncertainty. On the other hand, having all that data on how your operations are succeeding helps you improve them even more, and allows you more transparency in the eyes of authorities, partners and customers. Did you find this article useful? Take a look at our other blog megapost What is Data Logging? or the posts New EU Regulations Require QR Codes on Medicinal Packaging and Three Reasons Data Logging is Vital for Your Pharma Company for similar content!