In the post What is Data Logging? I mentioned that in 2017, the cold-chain logistics spending was globally over $13 billion for biopharmaceutical goods alone. Of that, at least 20% is estimated to be damaged during shipping, with other estimations ranging even up to 40%. With the global spending in the industry being in constant rise, the total amount of pharmaceutical losses worldwide is immense.
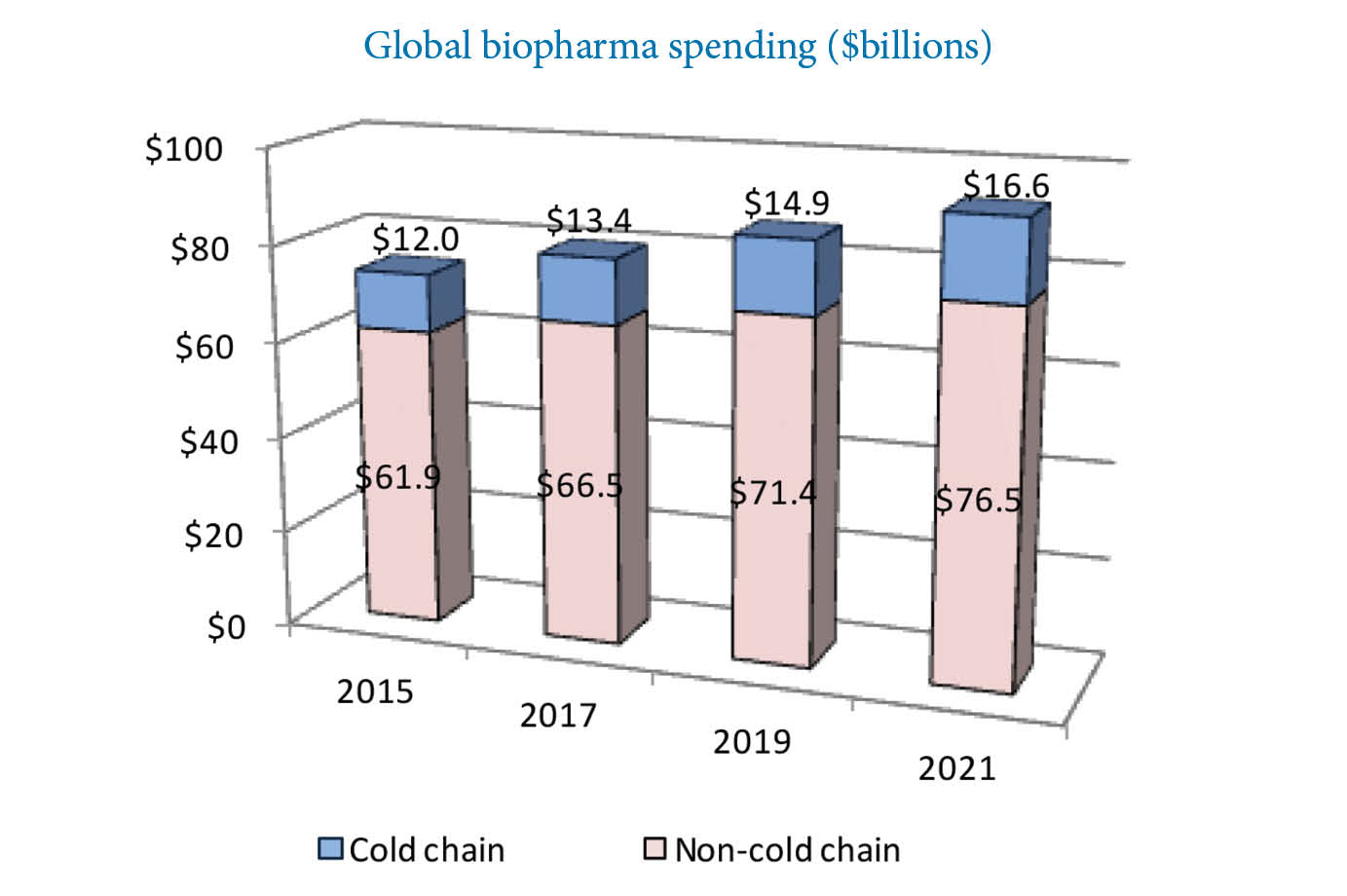 Global biopharma spending is continuously on the rise. Source: Pharmaceutical Commerce
Global biopharma spending is continuously on the rise. Source: Pharmaceutical Commerce
A 20% loss cannot be accepted
20% of all shipped goods being wasted is absolutely unacceptable, from both financial and the environmental standpoints, and 40% even less so. If you’re shipping a million’s worth of products every year, that 20% already means a fifth of a million’s worth of losses. At that point, it’d probably be better to just have 20% less business in general, since you would at least save the production costs. Now, let’s assume you could determine with higher precision whether a specific part of your shipment is still usable. How about being able to find out that on the average, only half of the contents of your ruined shipments is actually ruined? Your 20% loss is instantly halved. Assuming you’re shipping a million’s worth of products every year, that’s a 100 000€ saved with a simple investment on monitoring gear. Of course, the size of that investment is completely up to you, but the more precisely you can prove that a certain product is usable, the smaller the likelihood that you lose that product to mishandling. After all, simply having a visible means of monitoring is bound to encourage proper handling of the product.
Monitoring is required by the law
The World Health Organisation (WHO), the EU Commission, and even national legislative organs set various strict requirements to how pharmaceutical goods have to be handled during shipping and storing - and cold chain monitoring is one of the key aspects in this. In the US and Canada, the Centers for Disease Control and Prevention (CDC) and the British Columbia Centre for Disease Control (BCCDC) have set their own requirements and guidelines to how temperature-sensitive pharmaceutical goods should be handled. The different aspects that need to be considered by the manufacturer, retailers, and sometimes even the end-customer, include:
- Storage and handling methods for refrigerated and frozen vaccines
- Considerations for equipment such as storage units and thermometers
- Strategies for maintaining the cold chain
- Routine storage and handling practices
- Inventory management and emergency procedures for protecting vaccine inventories (Source: VBI Vaccines) The different guidelines set by these authorities exist to ensure that the pharma goods are safe to use - and effective.
Quality assurance increases trust
When you use a suitable data logger to monitor what your products go through during shipping and storing, you can not only be certain of the validity of your goods yourself, but also can also show the data to any authorities asking questions about your operations, and even to the end-users if the logger is exceptionally efficient (Logmore and Finnish pharmacies are running a pilot project, in which the data loggers are attached to the product until they reach the end-user - quality assurance to the very end!). The trust attained with transparent operations is what earns your company the credibility that secures you new partners and customers - and a good reputation all around.
In conclusion
Efficient data logging is vital to any cold chain dependent operations, or any temperature-sensitive logistics in general. While the price tag may sound like an additional expenditure at first, simply being sure of which products can actually be used and which not can lead to your bottom line increasing by a lot just because you get to minimize the amount of products you have to consider spoilt due to uncertainty. On the other hand, having all that data on how your operations are succeeding helps you improve them even more, and allows you more transparency in the eyes of authorities, partners and customers. I for one gladly spend 100€ today, if it means I’ll save 500€ tomorrow. Did you find this article useful? Take a look at our blog posts What is Data Logging?, Four Ways Technology Will Boost Your Warehouse Efficiency and Big Data in Supply Chains. To learn more about QR data loggers, take a look at the Logmore products.